|
True T/FĮnzymes can be denatured, meaning that they no longer work right. fever List some factors that can affect enzyme activity. True Enzymes work under a lock and key model in which the substrate is the key and the active site is the lock. 4 How many valence electrons does each carbon atom have? Its ability to bond with itself What gives carbon the ability to form chains that are almost unlimited in length? If it contains carbon (except CO2) What makes a substance organic? Are there any exceptions? Its Shape/Function which determines its active sites What determines the function of an enzyme? False-Įnzymes are proteins that act as catalysts which slow down chemical reactions. Nucleic Acids/Carry Genetic Information for Building Proteins/Nucleotides made up of 1 5 carbon sugar, 1 Phosphate, and 1 type of nitrogenous base List the 4 macromolecules of life, their functions and their monomers. 1.Carbohydrates/Readily Available source of energy/MonosaccharidesĢ.Lipids/Fats Stored energy/Glycerol and fatty acidsģ.Proteins/Building blocks of tissue and Enzymes (catalyst in chemical reactions)/Ĥ. any base Identify two solutions that have more OH- ions than H+ ions. saltwater any acid Identify two solutions that have more H+ ions than OH- ions. Blood solution Homogenous Mixture in which all substancesĮx. Water in Salt water Suspension Mixture of water and non dissolved substanceĮx. 
salt in saltwater solvent The thing doing the dissociatingĮx. cinnamon sugar Solute the thing being dissociatedĮx. Mixture Physical combination of two are more substancesĮx. False T/FĬapillary action only occurs in plants. True T/FĪ hydrogen bond is a weak bond between hydrogen and oxygen atoms. False T/FĬohesion is waters ability to stick to other carbon atoms. Waters ability to form solutions is due to its polarity. False T/FĪ hydrogen bond is stronger than a covalent bond. False T/FĬovalent bonds give water a low heat capacity. Hydrogen bonds are an example of adhesion. is the process that forms large organic molecules Polymerization When two or more.join together, a polymer forms monomers How did organic compounds get their name? How is the word related to its meaning? Organic means "living".Nucleic acid carbon compound that stores and transmits genetic information acid compound that forms hydrogen ions in solution nucleus the center of an atom isotopes atom of an element that differs in the number of neutrons compared with other atoms of the same element covalent bond formed when electrons are shared between atoms atom basic unit of matter polymer macromolecule formed when monomers join together True T/F Any amino acid can bond with any otherĪmino acids are assembled into polypeptide chains according to instructions coded in DNA Peptide bonds covalent bonds that link amino acids together to for a polypeptideĪmino acids are assembled into polypeptide chains according to instructions coded in DNA How many valence electrons does each carbon atom have? Each carbon atom has four valence electrons What gives carbon the ability to form chains that are almost unlimited in length? A carbon atom can bond to other carbon atoms Many of the molecules in living cells are so large they are called. More than 20 different amino acids are found in nature. Proteins control the rate of reactions, regulate cell processes, form cellular structures, carry substances into or out of cells, and help fight disease Amino acid has an amino group (-NH2) on one end and a carboxyl group (-COOH) on the other end. Proteins made up of nitrogen, carbon, hydrogen, and oxygen There are two kind of nucleic acids: DNA (deoxyribonucleic acid) and RNA (ribonucleic acid) Nucleotides has three parts: a 5-carbon sugar, a phosphate (-PO4) group, and nitrogenous base. 
Store and transmit hereditary (genetic) information. Steroids manufactured by the body are lipids as well Nucleic acids contain hydrogen, oxygen, nitrogen, carbon, and phosphorus 
They can be used to store energy and form parts of biological membranes and waterproof coverings Made mostly of carbon and hydrogen atoms. 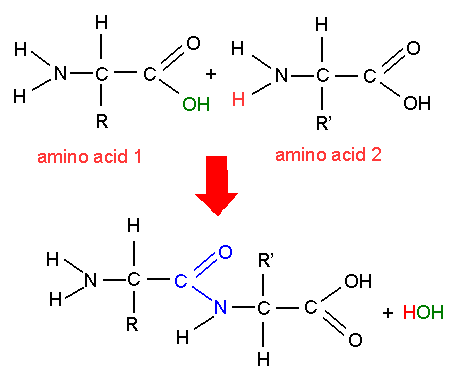
Plants and some animals also use carbohydrates for structural purposes Monosaccharides Molecules with one sugar monomer Disaccharide made of two monosaccharides Lipids fats, oils, and waxes They are the main energy source for living things Are composed of carbon, hydrogen, and oxygen
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
